By now, I’m sure you’ll know that I’ve been trekking around Sheffield’s nature reserves, collecting data for the National Lottery Heritage funded ‘Data 4 Nature’ project. The third and final set of surveys I carried out with ‘Data 4 Nature’ involved monitoring the condition of Sheffield’s woodland reserves. The Sheffield and Rotherham Wildlife Trust take care of some of the most beautiful woodlands in the area, including Wyming Brook: my favourite place to go take a breather from the stresses of university life. We are lucky to live in such a green city, and being 10.4% woodland by area, Sheffield is actually the most wooded city in Britain. We all know the benefits woodlands provide for our physical and mental health: they clean the air we breathe, give us space to exercise our bodies and reduce mental fatigue. We’re not the only ones to benefit though; woodlands are home to more wildlife than any other landscape. Take the humble oak tree – it supports birds nesting in its canopy, small mammals burrowing in its root system, bats roosting in its trunk and up to 280 different insect species living amongst its branches. We need to protect woodlands to make sure they continue to give wildlife a home. So, ‘Data 4 Nature’ has created a monitoring protocol to assess the condition of four key woodlands in Sheffield: Blacka Moor, Moss Valley, Fox Hagg, Wyming Brook. If we know the current state of our woodlands, management can be tailored to maximise benefits to wildlife in the future.
How do we define woodland condition?
‘Data 4 Nature’ developed a protocol for defining the condition of a woodland based on national standards. Woodland condition is broken down into two elements: structural attributes of the woodland, and species composition of the trees. When we talk about woodland structure, we are thinking about things like the amount of dead wood, how many glades are present, and how many invasive species there are in the area. Species composition considers which tree species are found in the woodland, as well as their diversity and age.
The woodland condition surveys started right back in autumn 2018. The team decided to do the surveys twice round, once in the winter, and once in the summer. The winter surveys focused on the structural aspects of woodland condition. We had to wait until the summer to look at species composition – it’s incredibly hard to ID tree species with no leaves! Luckily, the ‘Data 4 Nature’ team don’t expect us to be that good at species ID.

Our method:
Each woodland reserve was divided up into 1-hectare grid cells, measuring 100m2. In teams of three or four, volunteers followed a pre-planned route through the grid cells, taking measurements as we went. These routes didn’t always stick to the footpaths, so we had to scramble up and down the valley edges to stick as closely to our route as possible (I only fell over once, surprisingly). The biggest challenge we faced was not getting lost in the middle of the woods! So, what were we looking for as we orientated our way between brambles and through thickets?
Evidence of browsing –
Browsing by woodland mammals can severely damage woodland biomass. We assessed how much vegetation in each survey plot was damaged by deer and squirrel browsing, to see how much it impacts the ecosystem. The deer population is currently growing at Blacka Moor Reserve – notable by the number of droppings we found across the landscape! – so it is especially important to ensure deer are not stripping away too much of the woodland. We saw a herd of deer tracking though Blacka Moor during our survey. I’ve never seen deer out in the daylight before, so this was a rare and special sighting.
Invasive species –
We identified and estimated the percentage cover of invasive plant species within each grid cell. In the winter, the most common invasive species you will see are rhododendron and cherry laurel. These species are evergreens, thankfully making them easy to identify in the winter months.
Rhododendron can be identified by its dark glossy leaves, elliptical in shape and measuring around 22cm. These leaves grow in a whorl around a hard, light brown stem. The species has attractive pink to purple flowers located in the middle of the terminal whorl of each branch. Rhododendron is believed to have been introduced to the UK from Gibraltar in 1763. By 1893, it was being sold on London markets as a flowering pot plant. Rhododendron is incredibly difficult to eradicate from landscapes; its small seeds disperses rapidly via the wind and it can grow to fill any space available to it, either remaining as a small shrub, or growing to outcompete native vegetation. Mechanical methods of removing it are highly costly. It was estimated that £30 million would be needed to clear the species from the Snowdonia National Park alone.
Cherry laurel is another invasive species which is easily confused with rhododendron. It also has leathery, dark green, oblong leaves, but these are duller and smaller than rhododendron leaves. Cherry laurel also has erect racemes of white flowers that give rise to black cherry-like fruits. The species was planted in the UK in the 17th Century and was often used for hedging and game cover. Its abundant fruit makes it easy to disperse via birds. Cherry laurel casts a dense shade that excludes other species from accessing light resources. This means it can dominate the shrub layer and reduce diversity in the ecosystem.
Estimating how well established these two species are in Sheffield reserves will allow the Trust to develop management strategies that best prevent it from hindering the growth of native vegetation.
Woodland edge –
Transitions zones between one habitat and another are called ecotones. Ecotones are most valuable to wildlife if change is gradual between neighbouring habitats. For example, an edge which transitions from woodland, to shrub, to grassland is better at allowing wildlife to move through it than woodland which abruptly turns to grassland. We looked at whether the woodland edge measured at least 1.5 times the height of the nearest mature trees, to see if there is a wide edge between the woodland and neighbouring habitat in our woodland reserves.
Temporary open space –
The Wildlife Trust need to know how much temporary open space is present in the woodland reserves. These are areas in which trees could regenerate if we stopped cutting back vegetation and allowed natural ecological succession to play out. Areas where trees could regenerate include glades, rides, footpaths and areas of clear-felling. These areas had to be at least 10m in diameter and have less than 20% shrub/tree cover to be counted in our surveys.
Glades and rides are also valuable because they replicate woodland edge habitat inside wooded areas. This creates a ‘heterogenous’ habitat: a habitat with lots of different microhabitats within it. For example, a heterogenous woodland may have some closed canopy areas, open canopy areas, scrubby areas, grassy areas, wetland areas and more. This means a whole host of wildlife adapted to these different microhabitats can be supported. Having ecotones both between habitat types and within woodland is highly valuable because of the range of wildlife they let thrive. Ecotones are more valuable than many other woodland habitats, excluding veteran trees.
Wetland features –
This one was easy – we simply recorded the presence of any pools, ponds, rivers, brooks, streams or bogs in our grid cells. The Trust are aiming to have no net loss of wetland areas in their reserves, as wetland features provide a crucial microhabitat for aquatic wildlife.
Presence of dead wood –
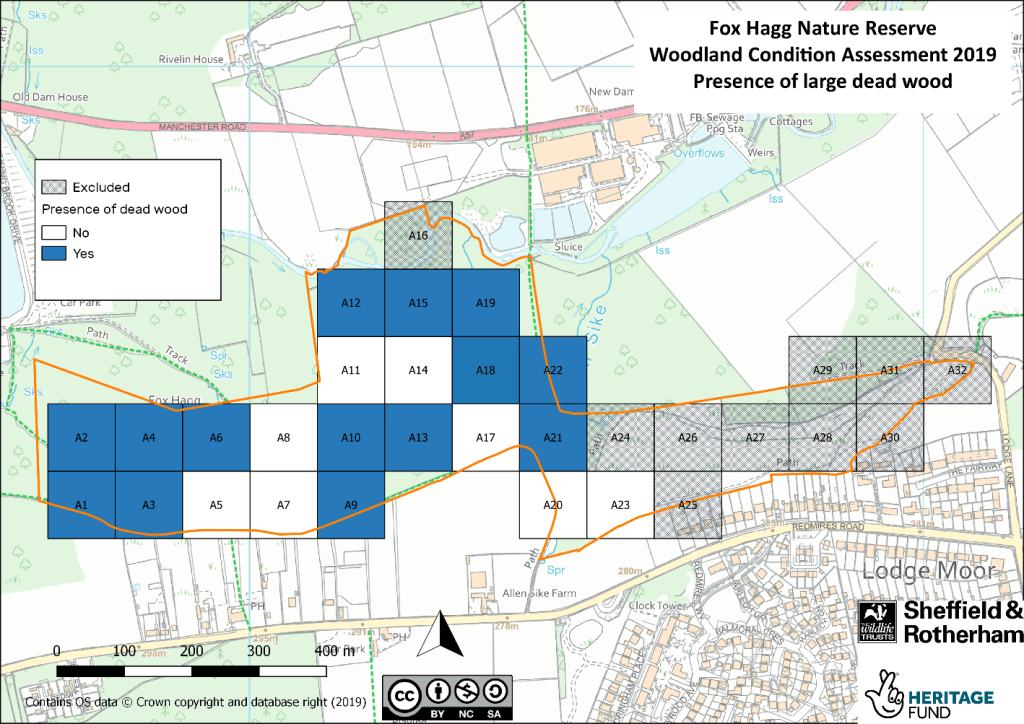
Identifying how much dead wood was present in each grid cell was important for the Trust to assess how suitable woodland areas are for Willow Tit populations: one of the fastest declining bird species in the UK. Willow Tits require open woodlands with scrubby areas and plenty of dead wood. The Trust have been working to increase the Willow Tit population in Fox Hagg over the past four to five years by providing more suitable habitat.
Species composition –
Once the days had started to draw out and the new spring leaves had burst-bud, we could assess the species composition aspect of woodland structure. We used ID sheets provided by the Trust to help us identify and record the presence of tree species in the woodland. There are 16 native and 7 non-native species that are common in the Sheffield area, which we encountered most regularly. We also put the trees into age classes based on the diameter of the tree at breast height. These age classes were: sapling, young, mature and very mature.
Results:
Overall, our surveys showed that the woodland reserves are in good condition. The results indicated that regeneration of native species is ongoing and species diversity was high across all sites. The Trust plan to use this data as a baseline to highlight any future changes in species diversity and composition in the woodland reserves.
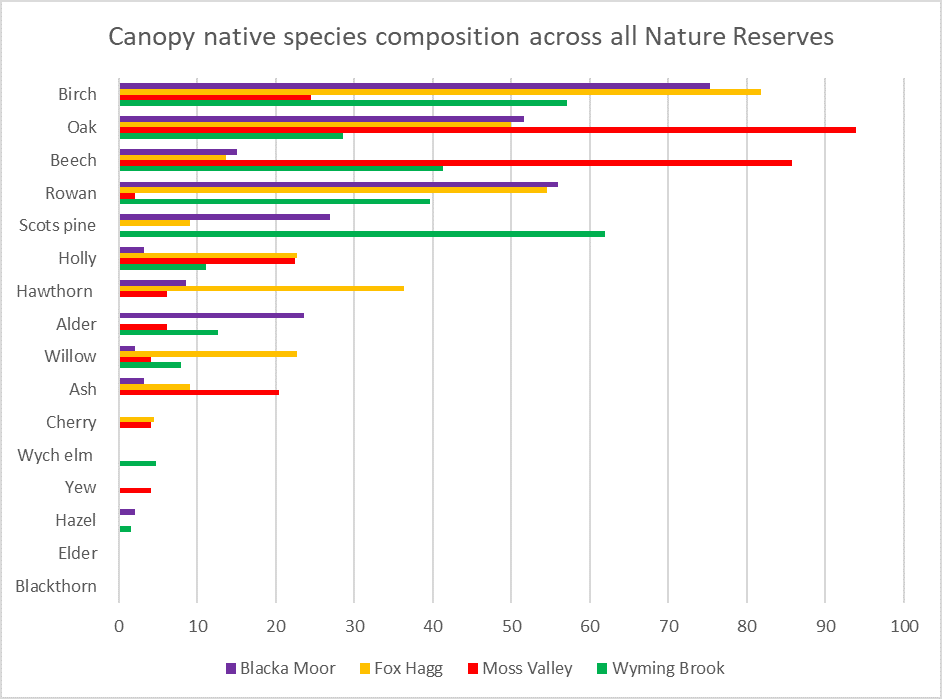
The Fox Hagg surveys showed that there is plenty of dead wood in the woodland to support a viable Willow Tit population. This is great news for the Land Management Team at Fox Hagg, who have been working to clear out glades for the species. However, Fox Hagg represents a difficulty that woodland managers often face: managing to benefit one particular species, versus managing the site as a whole. Normally, woodland managers would aim to have open space within 10-40% of grid cells. At Fox Hagg, open space is now found in 70% of grid cells to benefit the Williow Tit. This will make it less suitable for woodland inhabitants that prefer a closed canopy structure.
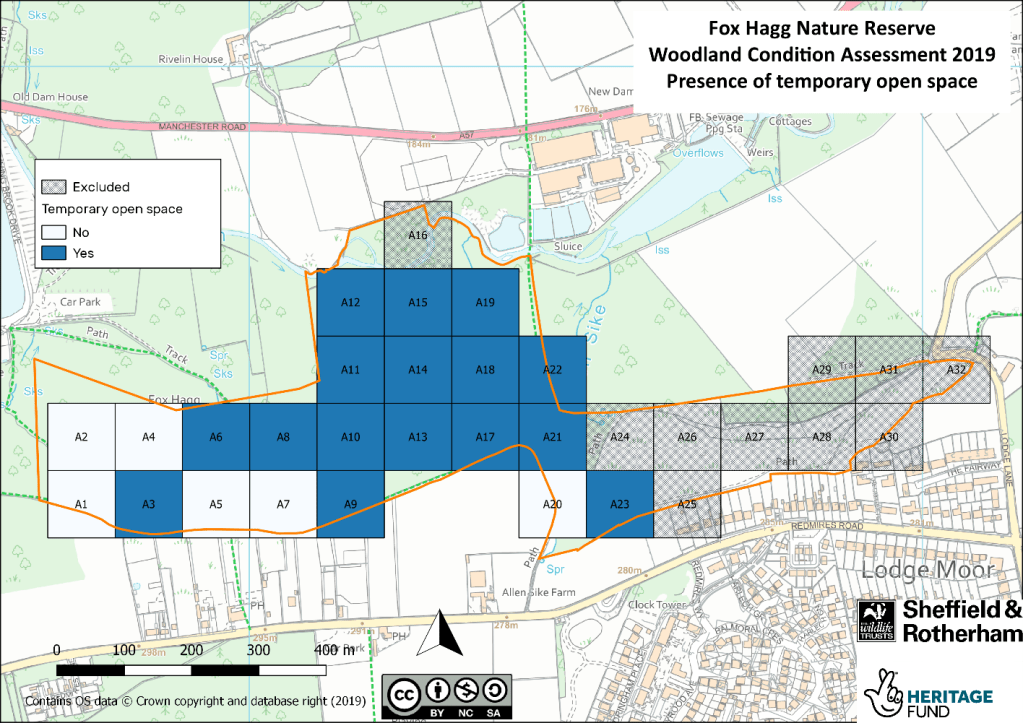
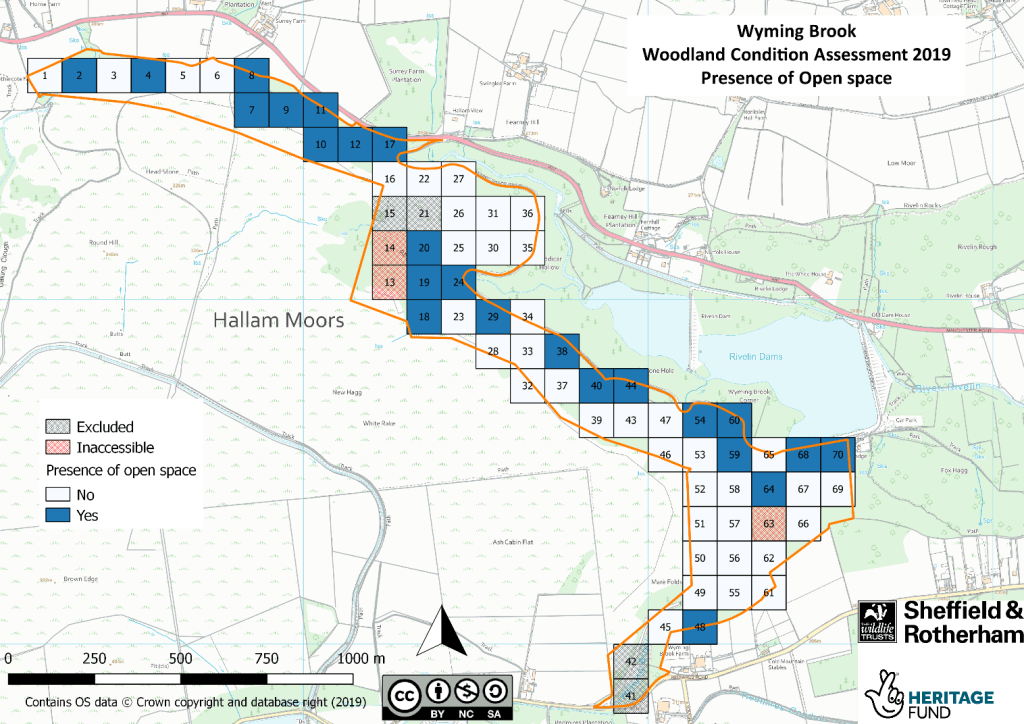
Wyming Brook, on the other hand, is an important habitat for the Wood Warbler: a species that prefers closed canopy mature woodland. Wyming Brook was found to only have open space in 40% of grid cells, showing it has been well-managed to support the Wood Warbler. Crucially, the data showed that grid cells without open space correlate with areas the Wood Warbler is known to occupy. This shows how the Trust can use the data to tailor management to each site specifically.
“The volunteers have done a fantastic job to collect and collate this monitoring data for Fox Hagg & Wyming Brook and it is very much appreciated. The data gives us a fantastic baseline to work with and it is great to see that we are meeting our targets for specific woodland condition attributes such as the presence of dead wood and temporary open space in the correct areas for our star species such as willow tit and wood warbler. The results will also help us identify areas where we can improve and target our management to help improve habitats for wildlife across these two fantastic nature reserves in the Rivelin Valley.”
Rob Miller, Reserve Manager
Volunteers spent over 500 hours rambling through Sheffield woodland to complete these surveys, so it’s great to see that the data we collected has already been used to improve management of woodland reserves. As always, I really enjoyed taking part in the woodland condition monitoring. It was a great opportunity to brush up on my tree species ID skills (crucial when you study ecology!), while exploring these beautiful woodlands. Get in touch with your local Wildlife Trust to find out how you can help protect wildlife in your area. Treat your mind and body to the benefits of being in woodland, whilst giving something back to the wildlife in your local area – what could be better?
- To find out more about why we should protect our woodlands, check out: https://www.woodlandtrust.org.uk/blog/2017/12/why-do-we-need-trees/
- You can learn more about the woodland reserves Sheffield and Rotherham Wildlife Trust look after here: https://www.wildsheffield.com/discover/nature-reserves/